Article Information
- Zhao Yang, Zhang Peng, Gang Hu Yi, Huang Lei . 2016.
- Effects of artificial vegetation arrangement and structure on the colonization and development of biological soil crusts
- Sciences in Cold and Arid Regions, 8(4): 343-349
- http://dx.doi.org/10.3724/SP.J.1226.2016.00343
Article History
- Received: February 26, 2016
- Accepted: April 28, 2016
Biological soil crusts (BSCs) are made up of cryptogamic species, such as cyanobacteria, green algae, lichen, moss and related microorganisms, which accounts for 40% of the living cover of desert ecosystems (West, 1990; Belnap and Lange, 2003). The ecological roles and ecosystem services of BSCs have been confirmed by a large number of reports and experiments (West, 1990; Belnap and Lange, 2003; Bowker, 2007; Li, 2012; Rozenstein and Karnieli, 2015).
In practice, BSCs are an important type of surface cover which occurs on the soil surface after their stabilization by re-vegetation (Li et al., 2007). Loss or degradation of BSCs also alters water redistribution (Eldridge et al., 2002; Li et al., 2010a, b), decreases fertility (Belnap and Lange, 2003), increases invasibility by exotics (Serpe et al., 2006), and, most importantly, increases soil erosion (Belnap and Lange, 2003; Li, 2012). Surface mining, such as coal mining, represents one of the most severe disturbances to forest and grass ecosystems in China, resulting in complete disruption of biotic and abiotic components of the ecosystem, especially the soil surface property of biotics such as BSCs (Brom et al., 2012). Therefore, the restoration of BSCs cover is necessary to these ecosystems. Li et al.(2010a)tested the relationship between plants and BSCs in the Tengger Desert, and highlighted that plant re-construction has created various habitats at a small-scale, affecting the colonization and development of BSCs. Zhao et al.(2010) examined the effects of different vegetation types (shrubs, semi-shrubs and trees) on the development of BSCs in Horqin Sand Land. They found that vegetation types have a significant impact on BSCs development, and documented that shrubs should be favored in future planting in arid and semi-arid regions. Zhao et al.(2016) also dominated that the effects of vegetation coverage on the colonization and development the BSCs should be considered in order to reconstruct the vegetation in disturbed environments. Therefore, re-vegetation ecosystems damaged by human activit y was shown to be an effective way to facilitate BSCs restoration. Typically, the vegetation landscape pattern in water-driven deserts is characterized by mosaic distributions of vascular plants and BSCs patches (Belnap and Lange, 2003). Therefore, it is difficult to identify the vegetation, particularly vegetation arrangement and structure such as coverage, height and density, as well as the effects on BSCs colonization and development in arid and semi-arid systems, and the results of recently conducted studies on their effects are still under debate (Bowker, 2007; Ochoa-Hueso et al., 2011; Li, 2012; Zhuang et al., 2015). Moreover, few studies have focused on the link between artificial vegetation and BSCs at dumping sites in surface coal mining areas, particularly those in arid and semi-arid regions.
In the present study, the coverage and thickness of BSCs, and coverage, height and density of woody vegetation were measured at a surface coal mine dumpsite to answer the following two questions:(1) Does vegetation arrangement and their structures strongly influence the colonization and development of BSCs?(2) Does the coverage and height of woody vegetation influence the restoration of BSCs?
2 Materials and methodsThis study was conducted at the north dumpsite (197 km2) of an opencast coal mine in Heidaigou (39°43′N-39°49′N, 111°13′E-111°20′E, average elevation: 1, 163 m), Junggar Banner, Inner Mongolia Autonomous Region, China. The annual average air temperature is 7.2 ℃, annual average rainfall is 426.3 mm, and relative humidity is 58%. Typically, the coverage and thickness of BSCs act as significant indices representing the colonization and development of BSCs (Belnap and Lange, 2003; Li, 2012). Therefore, the coverage and thickness of BSCs and coverage, height and density of woody vegetation were measured in the six re-vegetation types of Pinus tabulaeformis Carr.(PT), Populus alba var. pyramidalis Bunge (PA), Prunus sibirica L.(PR), P. alba var. pyramidalis Bunge, P. tabulaeformis Carr.(PPi), P. tabulaeformis Carr., P. sibirica L.(PPr), P. alba var. pyramidalis Bunge, P. tabulaeformis Carr., and P. sibirica L.(PPP), with no re-vegetated site as a control (CK). The descriptions of each site are presented in Table 1. At each vegetation type, three plots (10m×10m) were randomly chosen, and the coverage, height and density of woody vegetation were measured. An additional 10 plots (0.2m×0.2m) were randomly set up within each of the 10m×10m plots to measure the coverage and thickness of the BSCs. The coverage of the BSCs was measured by means of "Point Sampling Frame"(Li, 2005; Li et al., 2010a), and thickness was measured by Vernier caliper. Where ANOVAs show significant effects, Duncan's tests (P<0.05) were used to determine the differences. Next, regression analyses were used to examine the relationship between coverage, height and density of woody vegetation and coverage and thickness of BSCs. SPSS 16.0 software was used for statistical analysis.
| Re-vegetation types | Woody vegetationcoverage (%) | Woody vegetation height (%) | Woody vegetation density |
| Pinus tabulaeformis Carr.(PT) | 24.66±1.15 | 148.04±3.15 | 46.67±6.46 |
| Populus alba var. pyramidalis Bunge (PA) | 23.91±1.66 | 578.33±8.87 | 12.00±0.40 |
| Prunus sibirica L.(PR) | 2.27±0.12 | 73.01±3.11 | 14.70±1.52 |
| P. alba var. pyramidalis Bunge, P. tabulaeformis Carr.(PPi) | 17.45±1.27 | 565.65±4.18 | 15.30±0.82 |
| P. tabulaeformis Carr., P. sibirica L.(PPr) | 30.40±1.78 | 135.10±12.52 | 7.10±0.41 |
| P. alba var. pyramidalis Bunge, P. tabulaeformis Carr., P. sibirica L.(PPP) | 17.38±5.05 | 407.07±80.84 | 15.40±3.24 |
| Control (CK) | 0.00 | 0.00 | 0.00 |
There were significant differences among re-vegetation types in terms of the coverage of total BSCs, coverage of algae crust, coverage of moss crust and thickness of BSCs (P<0.05; Figure 1). PR, PT and PPr had the highest coverage of total BSCs, as much as 76.8%, 75.9% and 78.9%, respectively, which were significantly higher than PA, PPP and PPi which were 66.3%, 44.4% and 44.9%, respectively. The coverage of algae crust in PPr was 37.4%, which was also significantly higher than the other re-vegetation types. PR had the highest coverage of moss crust, as much as 60.5%, which was significantly higher than the other re-vegetation types. PR showed the thickest BSCs, at 4.41 mm, which was significantly higher than the other re-vegetation types.
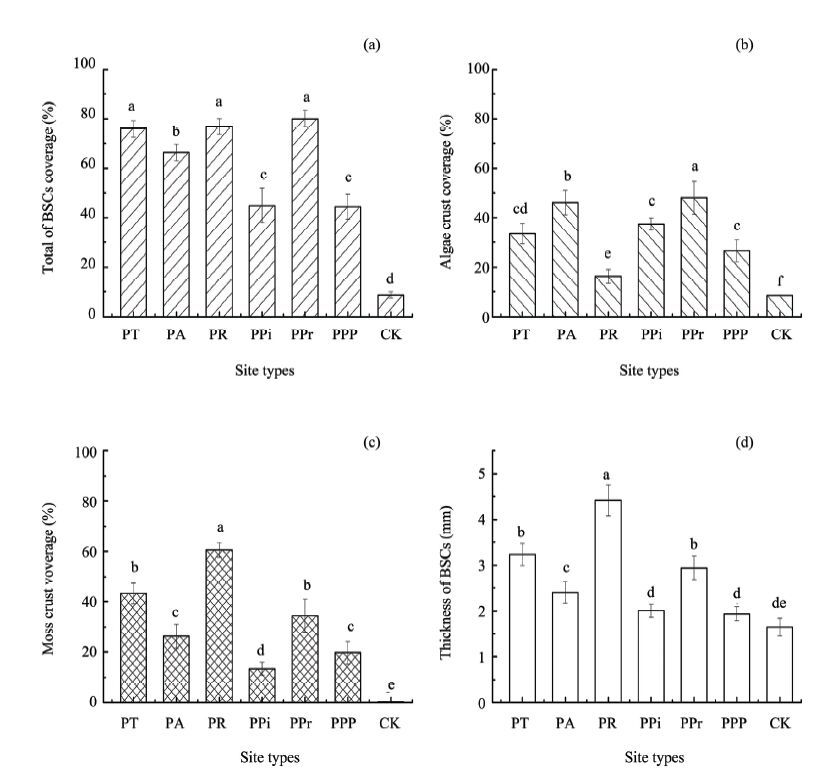
|
| Figure 1 Total coverage of BSCs (a), algea crust (b), moss crust (c) and thickness of BSCs (d) in the re-vegetation types of PT, PA, PR, PPi, PPr, PPP and CK |
There was a significant correlation between the coverage of total BSCs and coverage of woody vegetation (F = 47.802, P<0.001, df = 97; Figure 2a), as well as between coverage of algae crust and coverage of woody vegetation (F = 4.032, P = 0.021, df = 97; Figure 2b), and between coverage of moss crust and coverage of woody vegetation (F = 15.014, P<0.001, df = 97; Figure 2c).
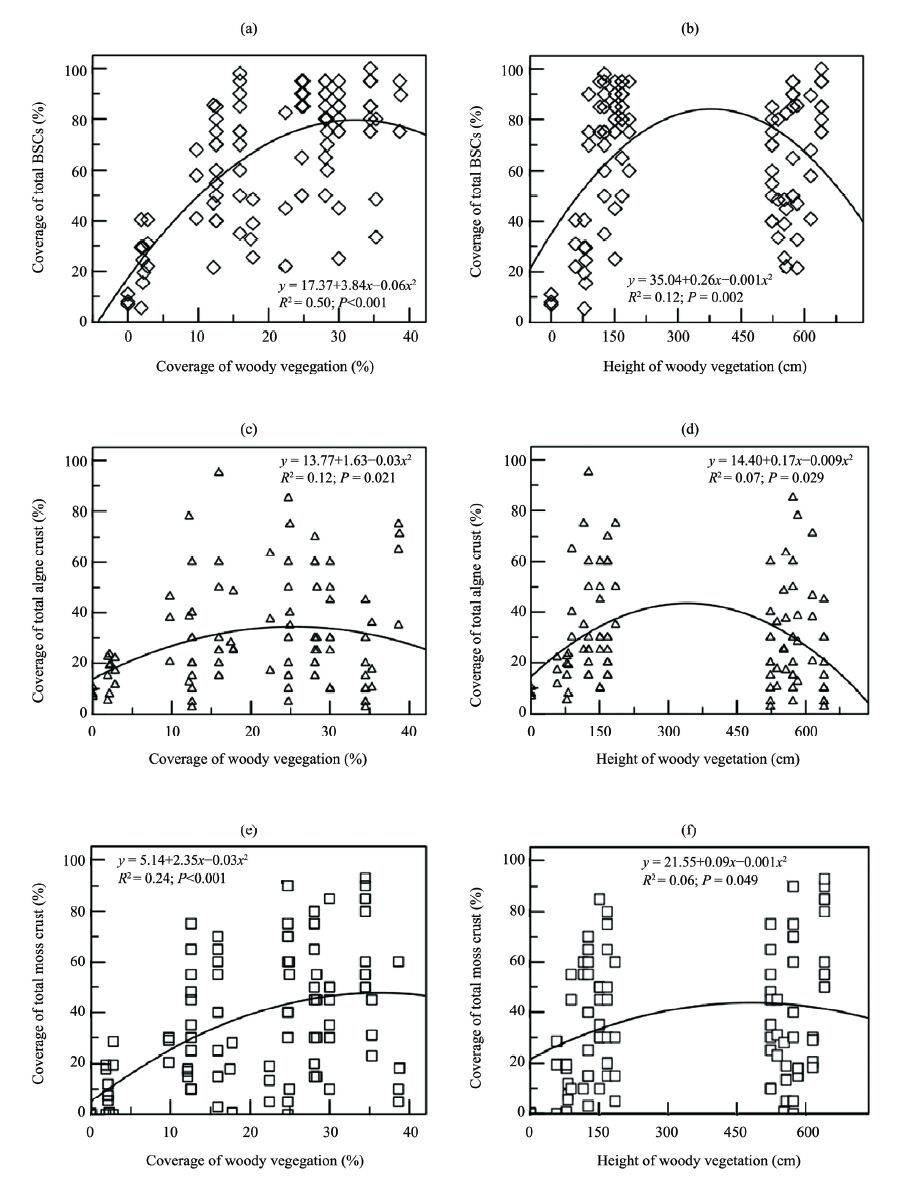
|
| Figure 2 Regression analyses of total coverage of BSCs and coverage of woody vegetation (a), and height of woody vegetation (b); coverage of algae crust and coverage of woody vegetation (c) and height of woody vegetation (d); coverage of moss crust and coverage of woody vegetation (e) and height of woody vegetation (f) |
There was also a significant correlation between the coverage of total BSCs and height of woody vegetation (F = 6.491, P = 0.002, df = 97; Figure 2d), between the coverage of algae crust and coverage of woody vegetation (F = 3.680, P = 0.029, df = 97; Figure 2e), and between the coverage of moss crust and coverage of woody vegetation (F = 3.563, P = 0.049, df = 97; Figure 2f). There was a significant correlation between the thickness of BSCs and coverage of woody vegetation (F = 3.121, P = 0.048, df = 97; Figure 3a), and height of woody vegetation (F = 3.182, P = 0.029, df = 97; Figure 3b) as a unimodal curve.
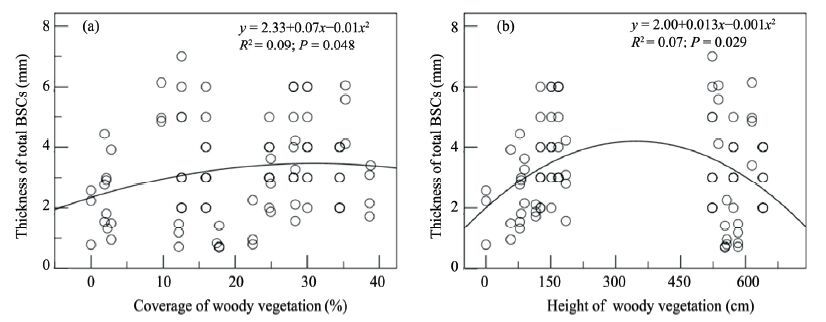
|
| Figure 3 Regression analyses of thickness of BSCs and coverage of woody vegetation (a), height of woody vegetation (b) |
Generally, shrubs have dense and limp branches and grow vigorously, thus they have a strong ability to decelerate wind speed and capture fine soil particles (Guo et al., 2008). On the other hand, trees, particularly larger tree species such as Populus alba var. pyramidalis Bunge, are fast-growing species and require substantial amounts of water and nutrients. Tree morphology may be the primary reason for their poor ability to reduce wind speed and control surface soil movement (Guo et al., 2008). Our results confirmed the results of a previous study by Zhao et al.(2010), which demonstrated that BSCs development was more prominent in shrub vegetation than in tree vegetation. Clearly, re-vegetation types and their distribution pattern significantly affect BSCs colonization and development. Therefore, shrub or shrub + dwarf tree vegetation types can facilitate BSCs restoration in the mine dumpsites in semi-arid regions. Our results also show that the coverage (total BSCs, algae crust and moss crust) and thickness of BSCs were higher in single woody plant species arrangement than double and trebling woody plant species arrangement. The main reason for this was that a greater number plant species living together leads to greater competition for resources such as water and nutrients, especially in ecosystems where water and nutrients are limited. Therefore, the planting of single woody plant species vegetation types can facilitate the restoration of BSCs in mine dumpsites in semi-arid regions.
Light is another vital factor on BSCs colonization. Strong light can inhibit the photosynthesis of BSCs and damage photoautotrophic organisms, thus reduced light intensity can be beneficial to BSCs colonization (Li, 2012). Typically, woody vegetation cover can screen direct solar radiation reaching to the soil surface, thus coverage by woody vegetation created suitable conditions for BSCs colonization. Guo et al.(2008)and Zhao et al.(2010) also reported that planted vegetation establishment was beneficial to BSCs colonization in semi-arid regions. However, complete shading by woody vegetation can also decrease the BSCs colonization rate (Schlensog et al., 2013; Zaady et al., 2013). Cryptogams are generally known as poor competitors for light in a plant community, and they show remarkable species-specific variation (Ochoa-Hueso et al., 2011). In the present study, the coverage of total BSCs, algae crust and moss crust decreased when the woody vegetation coverage reached more than 30% or 35%. Typically, light intensity decreases with an increase of woody vegetation coverage, in which case woody vegetation coverage is high enough to inhibit the photosynthetic activity of BSCs. Thus, BSCs colonization can be inhibited, which shows that the cover of BSCs was negatively associated with increasing woody vegetation coverage (Walker et al., 2006; Chaudhary et al., 2009; Ochoa-Hueso et al., 2011). This was also shown to be true in our study, demonstrating a negative correlation between the coverage of total BSCs and coverage of woody vegetation, as well as between the coverage of algae and moss crusts and coverage of woody vegetation in the late growth stage of re-vegetation.
In our study, shrubs and dwarf trees show a low level height, and larger trees show a high level height. Typically, shrubs and dwarf trees have a stronger ability to reduce wind speed than larger trees such as Populus alba var. pyramidalis Bunge (Guo et al., 2008). In addition, tree morphology may be the primary reason for their poor ability to reduce wind speed and control surface soil movement (Guo et al., 2008). Therefore, the height of woody vegetation significantly impacted the near-surface wind speed, which influenced the stability of the soil surface environment, and finally impacted the BSCs colonization. In the present study, the coverage of total BSCs, algae crust and moss crust were decreased when the woody vegetation height reached more than 300 cm. This implies that woody species with a height of no more than 300 cm should be selected for planting, as they are most suitable for BSCs restoration in mine dumpsites.
Our results show that correlation between the coverage of BSCs and coverage of woody vegetation is a unimodal curve. This implies that BSCs will rapidly colonize with an increase of woody vegetative coverage in the early stage of re-vegetation, but the BSCs colonization area will be destroyed and disappear with the further increase of woody vegetative coverage in the re-vegetation late stage. Therefore, woody vegetation coverage significantly affects BSCs colonization differently, depending on its growth stage. Land surface covered by woody vegetation and land near-surface wind speed will decelerate, and soil surface environment can subsequently be stabilized. Woody vegetation coverage can also screen direct solar radiation, reduce the daytime temperature of air and soil, and increase relative air humidity. As a result, the temperature and moisture regime can be stabilized on small and moderate scales (McPherson, 2007; Brom et al., 2012). A stable soil surface environment is a precondition for BSCs colonization (Belnap and Lange, 2003; Li, 2012). Before planting, the surface soil environment of the dumping site was unstable. By enhancing the soil surface roughness by vegetation with increase in roughness of near-surface (below 1 m), the wind velocity was significantly reduced (Li et al., 2004). This further increased dust falling from the atmosphere (d<0.063 mm) on the stable soil surface, and consequently the proportion of silt and clay in the soil surface layer was positively correlated with the accumulation of falling dust (Li et al., 2010a). The process eventually facilitated the colonization and growth of organisms of BSCs in the soil. The diurnal carbon fixation of BSCs determined the growth rate of the BSCs, which was largely regulated by available water and daytime moisture (Li, 2012). As poikilohydric, cryptogams are only metabolically and photosynthetically active when hydrated, and the persistence of their activity depends mainly on the site and form of hydration (Schlensog et al., 2013). In the present study, woody vegetation coverage increased the rates of transpiration through leaf cover, then subsequently increased the relative air humidity, and at the same time decreased air and soil temperature. In vegetation covered areas, as the vegetation coverage increases, relative air humidity increases and air and soil temperature decreases (Huang et al., 1987). An increase in relative air humidity can increase available water content, and a decrease in air and soil temperature can reduce the water evaporation from BSCs and prolong wet daytime. Therefore, woody vegetation establishment at the site was beneficial to the growth of BSCs.
Bowker (2007)highlighted the fact that the establishment of a stable soil surface, gentle light intensity, and available water and nutrients was a fundamental principle for BSCs restoration in practice. To a certain extent, the establishment of artificial vegetation can induce such conditions for BSCs re-colonization and development in degraded and damaged areas. However, once the woody plant coverage and height exceed the threshold level (in our case 30% and 300 cm, respectively), and the arrangement of woody plants involves more than two species, this can significantly negatively affect the BSCs colonization and development. These findings suggest that the influence of woody plant arrangement and structure on BSCs colonization and development should be considered in the re-construction of vegetation. Therefore, it is concluded that the coverage and height of woody plants should be controlled within the threshold level, in order to ensure sustainable colonization and development of BSCs in the process of vegetation restoration in semi-arid regions.
Acknowledgments:This work was sponsored by the National Natural Scientific Foundation of China (41501270 and 41530746) and Foundation for Excellent Youth Scholars of Cold and Arid Regions Environmental and Engineering Research Institute, Chinese Academy of Sciences.
| Belnap J, Lange OL, 2003. Biological Soil Crusts:Structure, Function, and Management. Berlin, Heidelberg: Spring-er-Verlag.. |
| Bowker MA, 2007. Biological soil crust rehabilitation in theory and practice:an underexploited opportunity. Restoration Ecology, 15(1): 13–23. doi: 10.1111/j.1526-100X.2006.00185.x |
| Brom J, Nedbal V, Procházka J, et al, 2012. Changes in vegetation cover, moisture properties and surface temperature of a brown coal dump from 1984 to 2009 using satellite data analysis. Ecological Engineering, 43: 45–52. doi: 10.1016/j.ecoleng.2011.03.001 |
| Chaudhary VB, Bowker MA, O'Dell TE, et al, 2009. Untangling the biological contributions to soil stability in semiarid shrublands. Ecological Applications, 19(1): 110–122. doi: 10.1890/07-2076.1 |
| Eldridge DJ, Zaady E, Shachack M, 2002. Microphytic crusts, shrub patches, and water harvesting in the Negev desert, the Shikim system. Landscape Ecology, 17(6): 587–597. doi: 10.1023/A:1021575503284 |
| Guo YR, Zhao HL, Zuo XA, et al, 2008. Biological soil crust development and its surface soil properties in the process of dune stabilization, Inner Mongolia, China. Environmental Ge-ology, 54(3): 653–662. doi: 10.1007/s00254-007-1130-y |
| Li XR, 2005. Influence of variation of soil spatial heterogeneity on vegetation restoration. Science in China (Series D:Earth Sci-ence), 48(11): 2020–2031. doi: 10.1360/04yd0139 |
| Li XR, 2012. Eco-hydrology of Biological Soil Crusts in Desert Regions of China. Beijing: Higher Education Press. |
| Li XR, He MZ, Zerbe S, et al, 2010a. Micro-geomorphology determines community structure of biological soil crusts at small scales. Earth Surface Processes and Landforms, 35: 932–940. doi: 10.1002/esp.1963 |
| Li XR, Kong DS, Tan HJ, et al, 2007. Changes in soil and vegetation following stabilisation of dunes in the southeastern fringe of the Tengger Desert, China. Plant and Soil, 300(1-2): 221–231. doi: 10.1007/s11104-007-9407-1 |
| Li XR, Tian F, Jia RL, et al, 2010b. Do biological soil crusts determine vegetation changes in sandy deserts? Implication for managing artificial vegetation. Hydrological Processes, 24(25): 3621–3630. doi: 10.1002/hyp.7791 |
| Li XR, Xiao HL, Zhang JG, et al, 2004. Long-term ecosystem effects of sand-binding vegetation in the Tengger Desert, Northern China. Restoration Ecology, 12(3): 376–390. doi: 10.1111/j.1061-2971.2004.00313.x |
| McPherson RA, 2007. A review of vegetation-atmosphere inter-actions and their influences on mesoscale phenomena. Progress in Physical Geography, 31(3): 261–285. doi: 10.1177/0309133307079055 |
| Ochoa-Hueso R, Hernandez RR, Pueyo JJ, et al, 2011. Spatial distribution and physiology of biological soil crusts from semi-arid central Spain are related to soil chemistry and shrub cover. Soil Biology and Biochemistry, 43(9): 1894–1901. doi: 10.1016/j.soilbio.2011.05.010 |
| Rozenstein O, Karnieli A, 2015. Identification and characterization of biological soil crusts in a sand dune desert environment across Israel-Egypt border using LWIR emittance spectrosco-py. Journal of Arid Environments, 112(Part A): 75–86. doi: 10.1016/j.jaridenv.2014.01.017 |
| Schlensog M, Green TGA, Schroeter B, 2013. Life form and water source interact to determine active time and environment in cryptogams, an example from the maritime Antarctic. Oecolo-gia, 173(1): 59–72. doi: 10.1007/s00442-013-2608-9 |
| Serpe MD, Orm JM, Barkes T, et al, 2006. Germination and seed water status of four grasses on moss-dominated biological soil crusts from arid lands. Plant Ecology, 185(3): 163–178. doi: 10.1007/s11258-005-9092-1 |
| Walker MD, Wahren CH, Hollister RD, et al, 2006. Plant com-munity responses to experimental warming across the tundra biome. PNAS, 103(5): 1342–1346. doi: 10.1073/pnas.0503198103 |
| Zaady E, Arbel S, Barkai D, et al, 2013. Long-term impact of agricultural practices on biological soil crusts and their hydro-logical processes in a semiarid landscape. Journal of Arid En-vironments, 90: 5–11. doi: 10.1016/j.jaridenv.2012.10.021 |
| Zhao HL, Guo YR, Zhou RL, et al, 2010. Biological soil crust and surface soil properties in different vegetation types of Horqin Sand Land, China. Catena, 82(2): 70–76. doi: 10.1016/j.catena.2010.05.002 |
| Zhao Y, Zhang P, Hu YG, et al, 2016. Effects of re-vegetation on herbaceous species composition and biological soil crusts development in a coal mine dumping site. Environmental Management, 57(2): 298–307. doi: 10.1007/s00267-015-0607-9 |
| Zhuang WW, Serpe M, Zhang YM, 2015. The effect of li-chen-dominated biological soil crusts on growth and physio-logical characteristics of three plant species in a temperate de-sert of northwest China. Plant Biology, 17(6): 1165–1175. doi: 10.1111/plb.12359 |
 2016, 8
2016, 8


