Article Information
- TianPeng Gao, Rui Guo, XiangWen Fang, ZhiGang Zhao, GuoHua Chang, YinQuan Chen, Qing Zhang. 2016.
- Effects on antioxidant enzyme activities and osmolytes in Halocnemum strobilaceum under salt stress
- Sciences in Cold and Arid Regions, 8(1): 65-71
- http://dx.doi.org/10.3724/SP.J.1226.2016.00065
Article History
- Received: June 15, 2015
- Accepted: September 7, 2015
2. State Key Laboratory of Grassland Agro-ecosystems, Institute of Arid Agroecology, School of Life Sciences, Lanzhou University, Lanzhou, Gansu 730000, China;
3. School of Chemistry and Environment Sciences, Lanzhou City University, Lanzhou, Gansu 730070, China
1 Introduction
There are two aspects that affect the growth of a plant. The first is the ion toxic effect to a plant caused by osmosis(Munns and Tester, 2008). Under the condition of salt stress,intracellular solute increases in order to improve salt tolerance of the plant and then the plant can maintain a certain osmotic force of cells by continuously absorbing water from outside. The osmotic adjustment substances include inorganic ions which come from outside and enter the cells(such as Na+,K+) and intracellular synthetic organic solutes such as Proline and Betaine(Liu et al., 2011; Yang et al., 2013). The second is oxidative damage to a plant(Jung,2004). Under the threat of an adverse situation,active oxygen accumulated within plant cells disrupt the balance between come into being and elimination of active oxygen during normal metabolism. In order to adapt to the environment,the plant will generate defense mechanisms,such as non-oxidizing enzyme system and antioxidants. It has become a measure of plant to resistance to salt,drought and destruction(Mittler,2002).
In studies on the salt tolerance of plants,research has shown that Si4+ can improve the plant's endurance towards biological and non-biological impact. For example,Si4+ can improve drought resistance of wheat,salt tolerance of corn and broomcorn, and insect-resistance of rice(Ma,2004). Some experiments have shown that when tomato and cucumber are under salt stress,Si4+ can increase the activity of Superoxide Dismutase(SOD),Catalase(CAT) and Glutathione Reductase(GR)(Liang et al., 2003; Al-aghabary et al., 2004). K+ plays an important role in absorption,transporting and maintaining enzyme activity during the process of plant photosynthesis(Miao et al., 2010). Moreover,K+ can increase crop yield. Therefore,K+ is one of many factors affecting the plant's adaptation to the environment,the lack of which will reduce mechanical stability and crop nutrition,while lowering the crops' resistance to pathogens(Pettigrew,2008). Under the situation of salt stress,exogenous Ca2+ can lower lipid membrane peroxidation and slow the damage of salt stress to plants by increasing activity of antioxidant enzymes in cucumber seedlings(Han et al., 2010). Ca2+ can obviously promote K+ absorption under salt stress,lower accumulation of Na+ and increase the ratio of K+/Na+. Increased K+ can be used as an effective osmotic regulator to maintain osmotic pressure.
H. strobilaceum(Pall.)Bieb.(Chenopodiaceae: Halocnemum Bieb.)is a halobiotic plant found in Gansu and Xinjiang of western China. It is a dominant species,usually growing in saline soil lowl and s,lakeside salt, and deserts. At present,there are few studies on the physiological mechanism of salt tolerant plants in western China. In this work,we extracted different saline solutions from the growth environment of H. strobilaceum in order to examine the salt tolerance mechanism of H. strobilaceum.
2 Material and methodsSeeds of H. strobilaceum were planted in a container with vermiculite and halite(3:1), and watered with nutrient water(1/2 Hoagl and )after them sprouting until seedlings grow into 4 cm.
NaCl solution and composite salt solution in concentrations of 0.0%,0.9%,2.7%, and 5.4% were used to irrigate the seedlings once they achieved a growth height of 4 cm,once every two days,respectively. Among them,composite salt was collected from the natural habitat of H. strobilaceum,the average content of soluble salt in soil of 0-30 cm is 15.68%±0.75% at weight ratio. The following indices were tested after 20 days.
In this study,we used the r and om sampling method. Three plants were arbitrarily chosen from their respective natural habitats in western China. Three 10 kg soil samples(0-30 cm)from each location were crushed and dried naturally,of which 5 kg soil sample from each location was mixed evenly.
Soil samples were immersed in distilled water for 24 hours and stirred once every two hours. Clear liquid and extract liquid were taken from the mixed solution three times. Crystals were separate out by heating and evaporation. Finally,organic matter and the color of the crystals were removed by adding hydrogen peroxide to achieve a composite salt. A series of composite salt solution and pure NaCl solution(solution concentration of 0.0%,0.9%,2.7%, and 5.4%)were made with distilled water for st and by,respectively.
The Ion absorption method was used to measure the type and content of soil composite salt(ACSC,1984).
Fresh weight of plants was determined after cleaning with distilled water and drying with filter paper. Dry weight of plants was determined after baking at 105 ℃ for 5 min and drying at 75 ℃. Succulent degree is calculated with the following formula: succulent degree= fresh weight/dry weight.
After treatment with NaCl and composite salt solutions(different gradient)for 30 days,plant samples were collected and cleaned with deionized water,dried and ashed at 550 ℃ with a muffle furnace until it became ash. The ash is dissolved by 75% nitric acid,then deionized water was added to reach a solution volume of 100 mL. The content of Na+ and K+ was measured by a flame atomic absorption spectrophotometer TAS-990F made by Beijing Purkinje General Instrument Co. Ltd..
Physiological indices of H. strobilaceum seedlings grown in different salt stress conditions for 30 days were measured. The contents of Malondialdehyde(MDA),Proline(PRO),Betaine(BET)are detected by the methods of Li et al.(1999),Davey et al.(2005) and Ábrahám et al.(2010),respectively. Enzyme activities were determined according to the methods of SOD(Beyer Jr. and Fridovich,1987),CAT(Chance and Maehly, 1955),POD(Shah et al., 2001). Each measurement was repeated three times and the average value was chosen.
Test results are analyzed with ANOVA method and the graphics are drawn with the Origin 8.0. The LSD Large Significant Difference Test was adopted.
3 Results 3.1 Cation ratio in the soil composite saltThe positive ion mass fraction ratio in the soil composite salt composed of Na+ : Ca2+ : K+ : Si4+ is 3.14 : 0.116 : 0.44 : 0.03,respectively.
3.2 The impact of different salt solution concentration on the growth of H. strobilaceum seedlingsDifferent salt solution treatments for 20 days obviously stimulated the growth of H. strobilaceum seedlings, and a strong promotion of growth for the soil salt treatment. Low concentrations of NaCl and composite salt are able to promote an increase in seedling dry weight,with composite salt having a greater stimulation effect than NaCl. With an increase of salt solution concentration,the dry weight of H. strobilaceum seedlings under composite salt stress is obviously higher than that of seedlings under NaCl solution stress. When the concentration of composite salt is below 2.7%,seedling dry weight significantly increases with the rise of salt solution concentration(p <0.05). When the concentration of NaCl solution is below 0.9%,seedling dry weight is higher than that in the control. When NaCl concentrations are larger than 2.7%,the dry weight of the treated seedlings shows an obvious downtrend(Table 1).
| NaCl (%) | Dry weight (mg) | Succulent degree (%) | Composite salt (%) | Dry weight (mg) | Succulent degree (%) |
| 0.0 | 120.21±0.005f | 4.65±0.010d | 0.0 | 120.21±0.005f | 4.65±0.010d |
| 0.9 | 220.15±0.010d | 6.09±0.010c | 0.9 | 330.12±0.020b | 6.26±0.050b |
| 2.7 | 290.17±0.010c | 6.24±0.020b | 2.7 | 430.33±0.570a | 6.93±0.020a |
| 5.4 | 151.66±0.020e | 6.18±0.020b | 5.4 | 220.33±0.570d | 6.74±0.050c |
| Note: Different small letters mean significantly different at P = 0.05. | |||||
H. strobilaceum has strong salt tolerance. Treatment at 0.0%-2.7% salt solution concentration significantly stimulates the growth of H. strobilaceum and causes a significant increase of dry weight and succulent degree. When NaCl solution concentrations are over 2.7%,succulent degree and water content decrease. Moreover,the chlorosis degree of assimilating branches of H. strobilaceum is enhanced in correlation with a decrease in the number of assimilating branches,but the dry weight is higher than the contrast. Composite salt is more conducive in maintaining water content of H. strobilaceum seedlings than NaCl. With an increase of composite salt concentration,succulent degree is higher than the contrast. When the concentration of composite salt solution is higher than 2.7% the succulent degree of H. strobilaceum decreases in correlation with an increase of this concentration. In addition,in any concentration,the water content of seedlings under composite salt treatment is higher than that under NaCl treatment(Table 1).
3.4 The impact of different salt solution concentrations on K+ and Na+ content of H. strobilaceum seedlingsAfter stress treatment of NaCl solution,Na+ content is higher than the contrast. With the rise of salt solution concentration,seedlings under composite salt solution treatment will absorb more Na+ than seedlings under NaCl solution treatment. When the concentration of two salt solutions is higher than 2.7%,Na+ concentration will double(Figure 1a). With an increase of NaCl solution concentration,K+ will decrease dramatically. When the NaCl solution concentration is 5.4%,K+ content will double compared to the salt stress treatment. When salt solution concentration is between 0.9%-5.4%,seedlings under composite salt treatment has a greater absorption capacity than under NaCl solution(Figure 1b). When the composite salt solution is less than 2.7%,the seedlings will absorb more K+ in correlation with an increase of salt content. Compared with NaCl,composite salt promotes absorption of K+,the content of which is almost double that of NaCl. Under NaCl stress,K+/Na+ will decrease with an increase of salinity. Also,seedlings under NaCl solution treatment shows a downtrend,which is greater than that of composite salt.
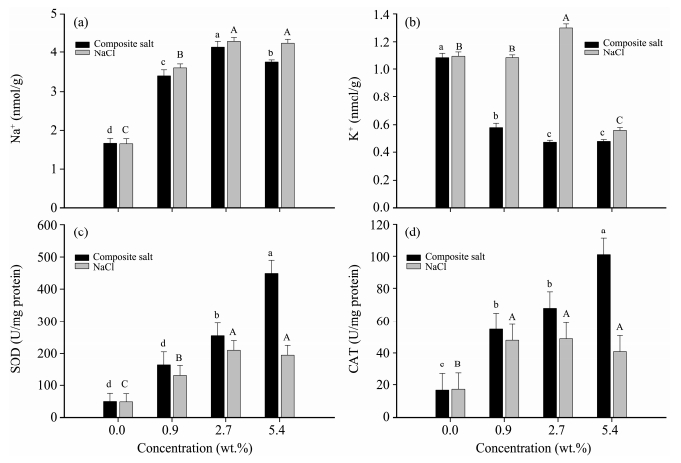 |
| Figure 1 The contents of Na+ (a), K+ (b), and the activities of SOD (c), CAT (d) of H. strobilaceum under treatments of NaCl and composite salts |
With an increase of salt solution concentration,SOD is higher than the contrast, and SOD activity of H. strobilaceum under composite salt treatment is higher than under NaCl treatment. Under composite salt treatment,an increase of salt solution concentration will strengthen SOD activity(Figure 1c). When the composite salt concentration is 5.4%,the maximum value of SOD activity is five times the contrast. Under NaCl treatment,the plant will show an uptrend with an increase of salt solution concentration,followed by a downtrend trend. SOD reaches a maximum value of 2.7% NaCl solution. In the NaCl solution with concentrations below 2.7%,H. strobilaceum can effectively decompose peroxide and reduce the harm to the cells. With a continuous increase of NaCl solution concentration,the ability of decomposing peroxide will gradually decrease.
3.6 CAT variation tendency under different salt treatmentsCAT variation tendency is basically the same as that of SOD(Figure 1d). SOD activity under composite salt treatment is larger than NaCl and will increase with the rise of salt solution concentration. When the composite salt concentration is 5.4%,the activity is 2.5 times of that under NaCl treatment. CAT can directly decompose H2O2 to H2O and O2 and effectively remove H2O2. As shown in the Figure 1d,CAT reached the maximum value in NaCl solution(2.7% concentration) and is about three times the contrast. A certain concentration of NaCl will be beneficial to the enhancement of CAT activity,but when the concentration is higher than 2.7%,CAT activity will decrease.
3.7 MDA variation tendency under different salt treatmentsAs the product of lipid peroxidation,the content of MDA can represent the degree of membrane damage. At normal growth conditions,the generation and removal of high concentrations of active oxygen within the plant are in balance. Under unfavorable stress,this balance is destroyed,which generates a large amount of MDA. As presented in Figure 2a,the variation trend of two salt solution treatments is similar and both show an upward trend. With an increase of salt concentration,MDA increases. When the solution concentration is 5.4%,MDA under composite salt treatment is 0.5 times of that under NaCl treatment(p <0.05),which shows strong membrane peroxidation.
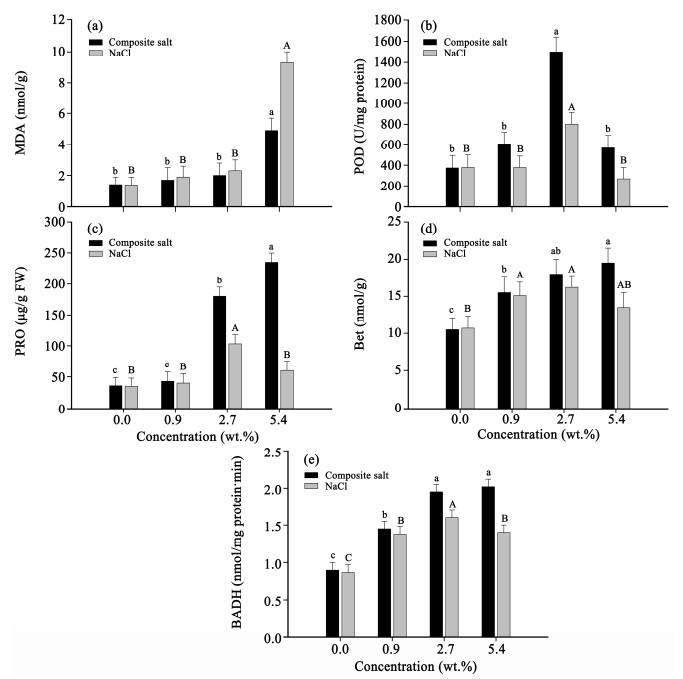 |
| Figure 2 The activity of POD (b), and the contents of MDA (a), PRO (c), Bet (d), and BADH (e) of H. strobilaceum under NaCl treatments and composite salts |
Through catalyzing H2O2 and reacting with the substrate,POD can consume H2O2 and reduce the damage of H2O2 to the organism. As presented in Figure 2b,when the salt solution concentration is less than 2.7%,POD activity is higher than the contrast. A certain concentration of salt solution promotes an increase of POD activity. Under composite salt treatment,POD activity is higher than that under NaCl treatment. When the concentration of both types of salt is 2.7%,POD reaches the maximum value. When the concentration of composite salt and NaCl is 5.4%,POD activity reduces. Compared with 2.7% salt concentration,POD activity decreases by 55% under composite salt treatment, and decreases by 63% under NaCl treatment. When the concentration of NaCl is 5.4%,POD activity is lower than the contrast. Thus,when the salt solution concentration is more than 2.7%,POD activity is inhibited,of which the influence of salt damage on the plant has surpassed its regulation capability. Compared with NaCl,the composite ion effectively regulates POD activity of H. strobilaceum in high concentrations,adapting to the growth environment of high salt concentrations.
3.9 PRO variation tendency under different salt treatmentsUnder the stimulus of composite salt,an increase of concentration will increase PRO content. When the salt concentration is 5.4%,the content of proline is 8.3 times of the contrast(Figure 2c). The composite ions effectively increase the content of proline under salt stress and relieve damage to the cells. Under NaCl treatment,low concentration salt solution is beneficial to the accumulation of proline. When solution concentration is larger than 2.7%,proline content will decrease but is still higher than the contrast. In the high concentration NaCl treatment(5.4%),proline content is 29% of that under composite salt treatment.
3.10 Betaine variation tendency under different salt treatmentsBetaine's accumulation in plant cells can increase the cells' ability of permeation and adjustment,stabilize intracellular macromolecular protein,biological membrane structure and function, and protect enzyme activity of many important metabolic activities. Under different concentration of NaCl and composite salt solution treatments,the betaine content is higher than the contrast. When NaCl solution is 2.7%,betaine content is the highest(Figure 2d). When NaCl solution concentration is more than 2.7%,betaine content will decrease with an increase of solution concentration. In composite salt treatment,betaine content will increase with an increase of salt concentration,reaching a maximum when the concentration is 5.4%. Irrespective of different treatments,betaine content is always higher than the contrast.
3.11 BADH variation tendency under different salt treatmentsThe activity of betaine aldehyde dehydrogenase(BADH)increases with a rise of composite salt solution concentration(Figure 2e). When NaCl solution is 2.7%,BADH activity reaches the maximum. The change of betaine content is consistent with the change of BADH. BADH activity of H. strobilaceum seedlings under composite salt treatment is higher than that of under NaCl solution treatment.
4 Discussion and conclusionsDifferent types of plants have different salt tolerances. Most plants with strong salt resistance have special structures,such as succulent stems and leaves of H. strobilaceum. Salt tolerant plants,on the one h and ,absorb salt ions, and on the other h and ,their leaves and stems become succulent in order to store a large quantity of water,dilute salt within the plant and maintain a certain level of salinity without affecting normal growth(Xu et al., 2000). This study shows that a salt solution concentration of 0.9%-5.4% promotes the degree of succulence in the leaves of H. strobilaceum,of which under composite salt solution stress is higher than that under NaCl treatment and the contrast. Si4+,K+,Na+ and Ca2+ of composite salt is more helpful in enhancing moisture holding capacity of H. strobilaceum than Na+ and NaCl. Previous studies have demonstrated that Si4+ of composite salt can reduce evapotranspiration and enhance water use efficiency(Gao et al., 2006).
According to MDA test results,under low concentrations of NaCl and composite salt solution treatments,MDA content varied little. Thus,a small increase of active oxygen produces a small impact on membrane lipid peroxidation. As a result,membrane structure is almost unharmed. Under high concentrations of NaCl treatment,MDA increases significantly,which is almost 7.75 times of the contrast. Thus,the more oxygen free radical(OFR)is accumulated,the greater the effect on membrane lipid peroxidation. As a result,cell membrane is damaged to some extent,but under the impact of composite particles,this damage changes slowly. Composite ions K+ and Si4+ are helpful in protecting cell membranes. Relevant research shows that under an environment with insufficient K+ or lack of K+,the membrane lipid peroxidation of plants is strengthened. After a supplement of K+,the membrane lipid peroxidation effect is obviously weakened(Hu et al., 2011).
The external ratio of K+/Na+ has some impact on the salt tolerance of plants. K+ can strengthen the salt tolerance of crops. Under salt stress,increase of medium K+ can reduce Na+ concentration of leaves. Moreover,an increase of K+ concentration can significantly slow down enhancement of membrane permeability caused by salt stress and increase the activity of root plasma membrane ATP enzyme. When the ratio of K+/Na+ is 0.9%,H. strobilaceum maintains a steady state of Na+ and K+. With the rise of salt concentration,K+/Na+ of H. strobilaceum seedlings under composite salt treatment is obviously higher than that under NaCl treatment. When salt solution concentration is 0.9%,K+ content of H. strobilaceum seedlings reached the maximum and K+/Na+ ratio is the highest. Meanwhile,existence of Ca2+ may enhance absorption of K+,lower accumulation of Na+ and increase K+/Na+ so as to lower and relieve the ion poisoning effect(Wang et al., 2009).
Among H. strobilaceum seedlings under composite salt treatment,the content of proline and betaine is higher. Ca2+ in composite salt may be helpful in increasing the content of proline in H. strobilaceum,which is consistent with Wang et al.(1999)observation that Ca2+ can increase proline content of Ficus carica. Our test results show that a high content of proline means high water retaining capacity and more effective antioxidant enzyme activity. High compound ion concentrations can promote synthesis of betaine. According to Ahmad and Haddad(2011),Si4+ of compound ions may increase the content of betaine. Meanwhile,an increase of betaine is helpful in enhancing the activity of antioxidant enzyme,reduces the damage of oxygen free radical(OFR)to plasma membrane and is beneficial to the stability and integrity of cell membranes.
There are numerous mechanisms for adapting to salt stress,including maintaining a high level of antioxidant capability or increasing activity of antioxidant enzyme. According to our test results,the activity of CAT,POD and SOD increased under low concentrations of NaCl treatment. Also,under the same conditions,the activity of CAT,POD and SOD under composite salt treatment is higher than that in NaCl treatment. Under salt stress,the plant will generate excess H2O2,which cause oxidation of cells. CAT can effectively decompose H2O2 to O2. Compound ions can effectively increase CAT content that effectively protects cell structure. Under composite salt treatment,CAT content will increase with the rise of salt concentration. In high concentrations of salt solution,compound ions are more helpful in increasing CAT content. Under low salt stress,POD accumulation shows an uptrend. By adjusting the activity of SOD,POD and CAT to enhance the resistance of salt stress,the free radicals of H. strobilaceum can maintain a relatively low level to prevent free radicals from damaging the membrane system and protect membrane structure. SOD is a kind of ubiquitous metalloenzyme in the biological body. Being promoted by compound ions,SOD content will increase with the rise of salt concentration. According to Guan(1999),Ca2+ and Si4+ can increase the activity of SOD in salt solution.
Synergistic effect of compound ions can effectively reduce the salt's toxic effect on H. strobilaceum. Ions such as Si4+,K+, and Ca2+ enhance the water retention capability of H. strobilaceum under high concentrations and promote the increase of succulence degree. Ions' toxic effect under low concentrations is harmful to H. strobilaceum. Through increasing absorption of K+,ions of Si4+,K+,Ca2+ can maintain a balance in the Na+/K+ ratio and effectively avoid the toxic effect of Na+,meanwhile,increase the organic solute content of proline and betaine to prevent the cells from being damaged and increase the biomass of H. strobilaceum. Moreover,synergistic effect of compound ions lowers the oxidation effect of plasma membrane under the effect of compound ions and increases the activity of antioxidant enzyme. This research provides a theoretical basis for halophytes to adapt to a saline environment and improve the plants' salt tolerance and output.
Acknowledgments: The authors thank Dr. Dan Su for her assistance with measurements at the experimental station. This work was funded by the National Natural Science Foundation of China(Grant Nos. 31160118,31460162,31422011,31370402, and 30800122).| Ábrahám E, Hourton-Cabassa C, Erdei L, et al., 2010. Methods for determination of proline in plants. Methods in Molecular Bi-ology, 639:317-331. DOI:10.1007/978-1-60761-702-0_20. |
| Agricultural Chemistry Specialty Committee of Soil Science Society Chinese (ACSC), 1984. Routine Analysis Method of Soil Agricultural Chemistry. Beijing:Science Press. |
| Ahmad ST, Haddad R, 2011. Study of silicon effects on antioxidant enzyme activities and osmotic adjustment of wheat under drought stress. Plant Breed, 47(1):17-27. |
| Al-aghabary K, Zhu ZJ, Shi QH, 2004. Influence of silicon supply on chlorophy II content, chlorophy II fluorescence and anti-oxidative enzyme activities in tomato plants under salt stress. Journal of Plant Nutrition, 27(12):2101-2115. DOI:10.1081/PLN-200034641. |
| Beyer Jr. WF, Fridovich I, 1987. Assaying for superoxide dismu-tase activity:some large consequences of minor changes in conditions. Analytical and Bioanalytical Chemistry, 161(2):559-566. |
| Chance B, Maehly AC, 1955. Assay of catalases and peroxidases. Methods in Enzymology, 2:764-775. DOI:10.1016/S007 6-6879(55)02300-8. |
| Davey MW, Stals E, Panis B, et al., 2005. High-throughput deter-mination of malondialdehyde in plant tissues. Analytical Bio-chemistry, 347(2):201-207. DOI:10.1016/j.ab.2005.09.041. |
| Gao XP, Zou CQ, Wang LJ, et al., 2006. Silicon decreases transpi-ration rate and conductance from stomata of maize plants. Plant of Plant Nutrition, 29(9):1637-1647. DOI:10.1080/01904160600851494. |
| Guan JF, 1999. Effects of calcium ion on cell membrane per-meability, activities of protective enzymes and content of protective substances in apple fruits. Chinese Bulletin of Bo-tany, 16(1):72-74. |
| Han B, Sun J, Guo SR, et al., 2010. Effects of calcium on antioxi-dant enzymes activities of cucumber seedlings under salt stress. Acta Horticulturae Sinica, 37(12):1937-1943. |
| Hu W, Li JZ, Zhang SX, 2011. Effect of potassium on physiological indices of tomato cultivars with different resistances to ne-matode. Chinese Journal of Eco-Agriculture, 19(1):98-102. DOI:10.3724/SP.J.1011.2011.00098. |
| Jung S, 2004. Variation in antioxidant metabolism of young and mature leaves of Arabidopsis thaliana subjected to drought. Plant Science, 166(2):459-466. DOI:10.1016/j.plantsci. 2003.10.012. |
| Li YQ, Gao JH, Zhou WR, et al., 1999. Determination of betaine content in synthetic betaine product by colorimetric method. Jiangsu Journal of Agricultue Science, 2:64-65. |
| Liang YC, Chen Q, Liu Q, et al., 2003. Exogenous silicon (Si) increases antioxidant enzyme activity and reduces lipid peroxidation in roots of salt-stressed barely (Hordeum vulgare L.). Journal of Plant Physiology, 160(10):1157-1164. DOI:10.1078/0176-1617-01065. |
| Liu HY, Cui CH, Zhao Q, et al., 2011. Effects of organic fertilizer on growth and endogenous hormone contents of wheat seedlings under salt stress. Acta Ecologica Sinica, 31(15):4215-4224. |
| Ma JF, 2004. Role of silicon in enhancing the resistance of plant to biotic and abiotic stress. Soil Science and Plant Nutrition, 50(1):11-18. DOI:10.1080/00380768.2004.10408447. |
| Miao BH, Han XG, Zhang WH, 2010. The ameliorative effect of silicon on soybean seedlings grown in potassium-deficient medium. Annals of Botany, 105(6):967-973. DOI:10.1093/aob/mcq063. |
| Mittler R, 2002. Oxidative stress, antioxidants and stress tolerance. Trends in Plant Science, 7(9):405-410. DOI:10.1016/S1360-1385(02)02312-9. |
| Munns R, Tester M, 2008. Mechanisms of salintiy tolerance. Annual Review of Plant Biology, 59:651-681. DOI:10.1146/annurev.arplant.59.032607.092911. |
| Pettigrew WT, 2008. Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiologia Plantarum, 133(4):670-681. DOI:10.1111/j.1399-3054.2008. 01073.x. |
| Shah K, Kuma RG, Verma S, et al., 2001. Effect of cadmium on lipid peroxi-dation, superoxide anion generation and activities antioxidant enzymes in growing rice seedlings. Plant Science, 161(6):1135-1144. DOI:10.1016/S0168-9452(01)00517-9. |
| Wang LJ, Liu YL, Ma K, 1999. The role of calcium in promotion of proline accumulation induced by salt stress in Fig (Ficus carica L.) cells. Plant Physiology Journal, 25(1):38-42. |
| Wang ZQ, Wang FF, Lin TB, 2009. Effects of calcium on proline content and related enzymes activities in wheat seedlings under salinity. Journal of Henan Agricultural University, 43(5):475-479. |
| Xu XM, Yu HC, Li GF, 2000. Progress in research of plant tolerance to saline stress. Chinese Journal of Applied and Environmental Biology, 6(4):379-387. |
| Yang Y, Yang Y, Wang GX, et al., 2013. Antioxidative responses of Abies fabri seedlings to litter addition and temperature eleva-tion. Acta Ecologica Sinica, 33(1):53-61. DOI:10.5846/stxb201111021649. |
 2016, 8
2016, 8


